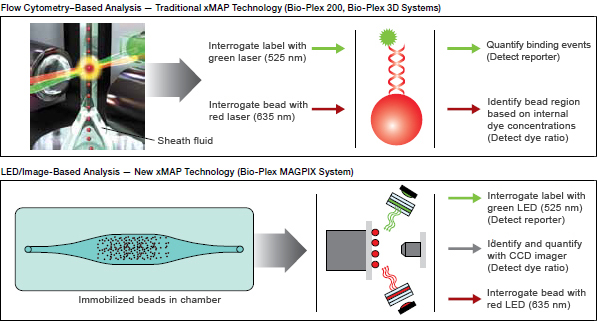
Recombinant GLURP-R0 and GLURP-R2 were expressed in E. coli, and EBA-175 was expressed in Pichia pastoris, and provided by David Narum, National Institutes of Health. Recombinant MSP-1 42 and MSP-3 were expressed in E. Lanar, Walter Reed Army Institute for Research. Recombinant AMA-1, LSA-NRC and TRAP were expressed in Escherichia coli and provided by David E. falciparum antigens used for testing were apical membrane antigen-1 (AMA-1, full length ectodomain FVO strain) erythrocyte-binding antigen (EBA-175, non-glycosylated region II) glutamate rich protein (GLURP, conserved non-repeat N-terminal region, amino acids 25–514, R0 and repeat C-terminal region, amino acids 705–1178, R2, 3D7 strain) liver stage antigen (LSA-NRC, C-terminal region, amino acids 1628 to 1909, 3D7 strain) merozoite surface protein-1 (MSP-1 19, E-KNG variant MSP-1 42 FVO strain) merozoite surface protein-3 (MSP-3, C-terminus, FVO strain) and recombinant thrombospondin-related adhesion protein (TRAP, 3D7 strain). falciparum recombinant and peptide antigens Ethical approval was obtained from Kenya Medical Research Institute National Ethical Review Committee (protocol number 2101) and the Institutional Review Board of University of Minnesota. Written informed consent was obtained from the study participants or, in the case of minors, from their parent or guardian. In addition, comparison of freshly coupled and 18-months stored coupled beads was performed by use of Bio-Plex 200. To compare the instrument detection limit (IDL) between the bead types 8 duplicate serially diluted positive control plasma at the following dilutions: 1: 50, 1:100, 1:200, 1:400, 1:1,000, 1:2,000, and 1:4,000 were analyzed by a 5-plex assay-(AMA-1, EBA-175, MSP-1(19), MSP- 1(42) and MSP-3). Comparisons of both coupled bead types were performed using the same plasma samples. To assess the utility of the coupled beads, a broad range of plasma test samples from 32 individuals from a malaria endemic area of western Kenya ( John et al., 2005) were also used. Additional similarities and differences between the two bead types are as shown in Table 1.įor all multiplex bead assays, a plasma pool made of 30 plasma samples from adults living in a Ugandan area of seasonal malaria transmission (positive control pool samples Idro et al., 2005) while seven plasma samples from malaria-naive North American individuals were used as negative controls. Magnetic beads are made of a core of magnetic material consisting of iron oxide and an internal array of dyes which color code the beads, thus allowing multiplexing ( Verkaik et al., 2008 Dunbar & Li, 2010 Houser, 2012). Recently, magnetic microspheres (beads) have become available as an alternative option to non-magnetic beads. These assays utilize microsphere beads, which are conjugated to specific capture reagents, including conjugation-ready bead sets carrying either avidin, carboxylate groups (COOH) or oligonucleotide adapters ( Reslova et al., 2017 Elshal & McCoy, 2006). On this front, there has been a surge in the development of multiplex methodologies as reliable replacements of ELISAs ( Cham et al., 2008 Lal et al., 2005 Verkaik et al., 2008). The capacity to measure relative levels of antibodies to multiple antigens could ultimately increase uniformity for comparative reasons. falciparum in comparison to a traditional ELISA (for example, sample requirements and handling). In many aspects, multiplex fluorescent microsphere assays are a more efficient method to detect plasma antibodies to P. Among the possible reasons for the inconsistent findings are differences in standardization and analytical approaches ( Fowkes et al., 2010). However, such studies have often yielded conflicting results with some demonstrating a protective effect for antibodies to a specific antigen ( Al-Yaman et al., 1996 Ogutu et al., 2009), while others do not show any association with malaria disease morbidity indicators ( Murungi et al., 2013 Fowkes et al., 2010). Cross-sectional studies of antibodies to one or two antigens have been used as markers to estimate past malaria exposure ( Cook et al., 2012 Cook et al., 2010 Drakeley et al., 2005). Antibodies to different Plasmodium falciparum antigens have been correlated in multiple immuno-epidemiological studies with protection from infection ( Duarte et al., 2012 Dent et al., 2012) or disease ( Lourembam & Baruah, 2012 Dodoo et al., 2011 Reiling et al., 2010).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
